
Since the accuracy in velocity is 0.005%, uncertainty in velocity, Δv = 600 x 0.005 / 100 = 3 x 10 -2 m s -1.ġ) State Heisenberg's uncertainty principle. Certainty with which the position of the electron can be located is: (h=6.6 x 10 -34 kg m 2 s -1 mass of electron, e m = 9.1 x 10 -31 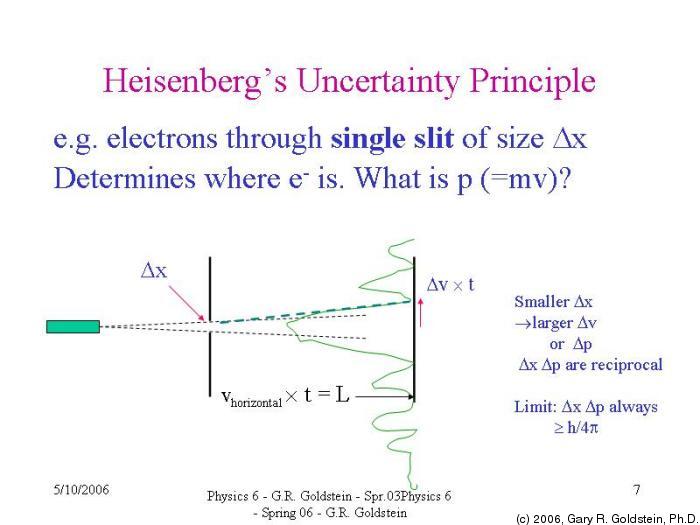
4) In an atom, an electron is moving with a speed of 600 m/s with an accuracy of 0.005%. The uncertainty in velocity, Δv is given by the expression:Ĭorrect option is "a". Hence, uncertainty in velocity (m s -1) is: ( Planck’s constant, h = 6.6 x 10 -34 J s) 3) Uncertainty in position of a particle of 25 g in space is 10 -5 m. What is the ratio of uncertainties?Ĭorrect option is "a". The Heisenberg uncertainty principle states that it is impossible to know simultaneously the exact position and momentum of a particle that is the more. The mass of B is five times to that of mass A. 2) The uncertainties in the velocities of two particles A and B are 0.05 and 0.02 m s -1 respectively. Note: h/n π is constant and Δv values are same.Ĭorrect option is "c". The ratio of uncertainties in positions for A and B is: The uncertainty in position, Δx is given by the expression: According to Heisenbergs uncertainty principle, the product of uncertainties in position (x) and velocity (v) is always equal to or greater than h/n. Related questions 1) If the uncertainties in velocities of two particles A and B with mass 1.0 x 10 -27 kg and 1.0 x 10 -31 kg respectively are same, the ratio of uncertainties in the positions of A and B is: The uncertainty in position (Δx) can be calculated as follows: It indicates that the uncertainty in velocity is 0.001% of actual value, 300 m s -1. What is the meaning of "accurate up to 0.001%"? IIT JEE - NEET Question: Uncertainty in the position of an electron (mass = 9.1 x 10 -34 kg) moving with a velocity 300 m s -1, accurate up to 0.001%, will be: (h= 6.63 x 10 -34 J s) (AIEEE 2006)Īccording to Heisenberg's uncertainty principle, the product of uncertainties in position (Δx) and velocity (Δv) is always equal to or greater than h/n π. UNCERTAINTY IN POSITION - VELOCITY - MOMENTUM Let's say you have a professional camera, and you want to take pictures of a tennis ball moving.Uncertainty in position | momentum | IIT JEE | Adichemistry Heisenbergs Uncertainty principle states that both the location (or position) and momentum of a particle can not be simultaneously ascertained accurately.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
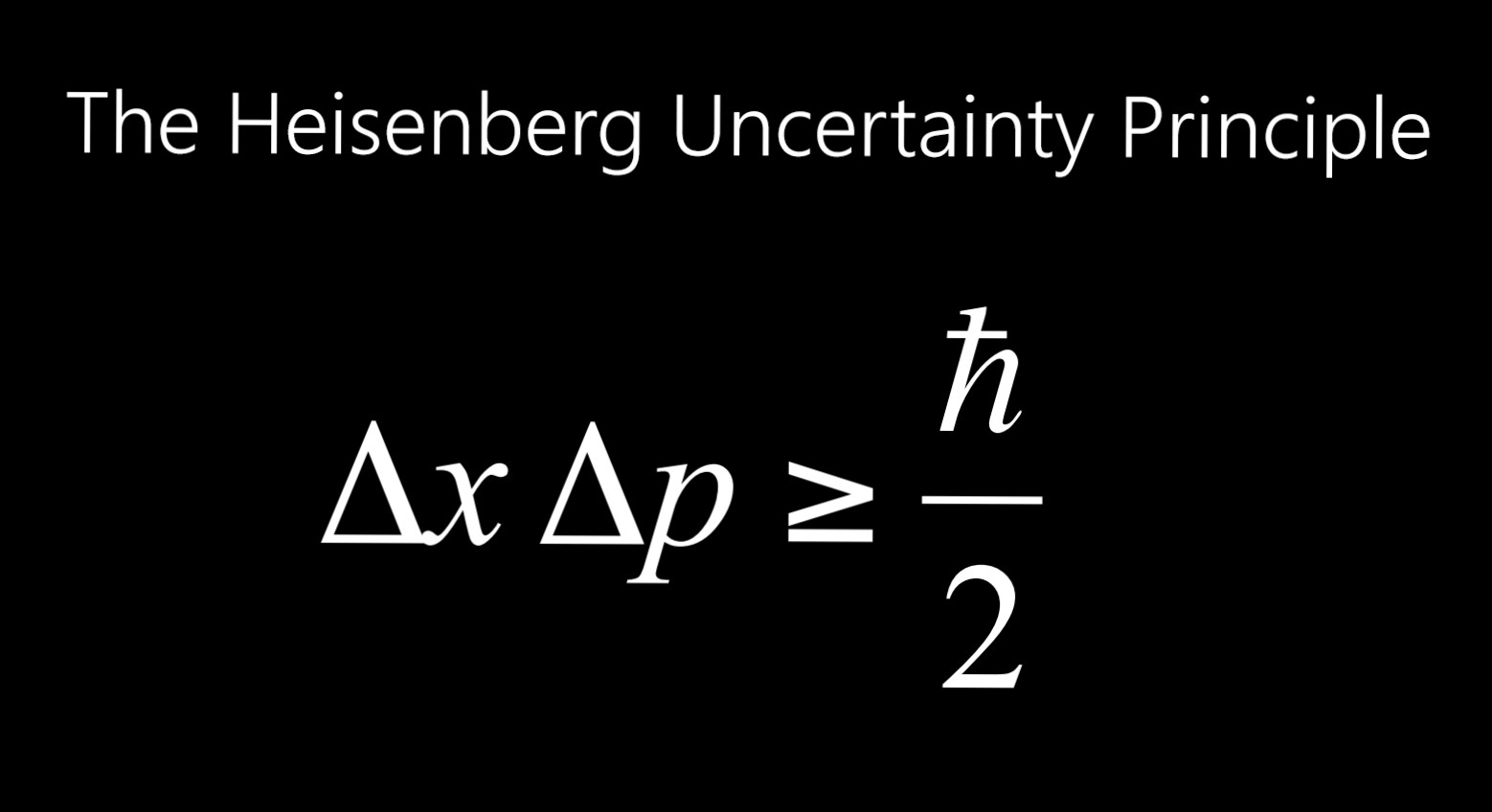
 RSS Feed
RSS Feed
